Alpha radiation: The particles we get from Alpha radiation are called Alpha particles. Alpha particles have energies of about 5MeV. This energy can ionize a plethora of atoms before these particles lose all their KE, because only about 10 eV is only needed to be ionize. This makes them harmful because if human were to encounter Alpha particles, their tissues would be damage, which will make their skin burn. However, because of their high reactivity and ionization, they only have a range of a few centimeters in the air and cannot penetrate paper. Alpha particles have the same energy. When a nucleus emits an alpha particle, 2 protons and 2 neutrons are lost.
Beta radiation: Beta minus particles are what we get from Beta radiation. They are exactly the same as electrons, but they are formed when a neutron changes to a proton. During Beta decay, a neutron changes into a proton and an electron. These particles travel farther with higher speed than Alpha particles, but they are not as ionizing and reactive as them. Since they are not as ionizing and reactive as Alpha particles, making them able to pass through more matter and more distance in the air. Beta particles does not have the same energy. When a nucleus emits beta particle, 1 neutron and proton are lost.
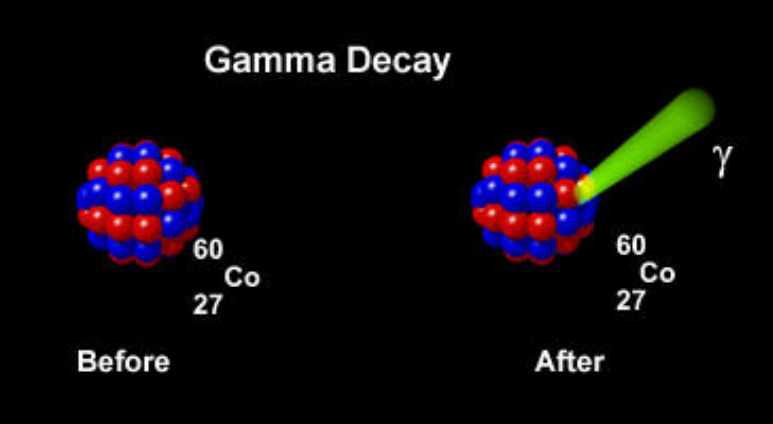
Gamma radiation: Gamma radiation creates no particles, instead it creates rays that are similar to X-ray. In a gamma radiation there is no change in the nucleon, but the emitted energy is just lost. Knowing that per nucleus decay emits photon, gamma radiation also emits photon. (Gamma photon are often emitted when a nucleus is left in an excited state after emitting another form of radiation). Gamma photon has very high energy, which are similar to the energy of a nuclear explosion. Their high energy allows them to ionize when they are absorbed by atomic electrons. Gamma radiation are what created the Hulk, so don't play with it :)
Radioactive Decay Equation